 How do you write the electron configuration for Uranium? What is the electronic configuration of Uranium 92? What is the boiling Point of Uranium in Kelvin?īoiling Point of Uranium in Kelvin is 4200 K. Melting Point of Uranium in Kelvin is 1408 K. What is the melting Point of Uranium in Kelvin? What is the boiling Point of Uranium?īoiling Point of Uranium is 4200 K. 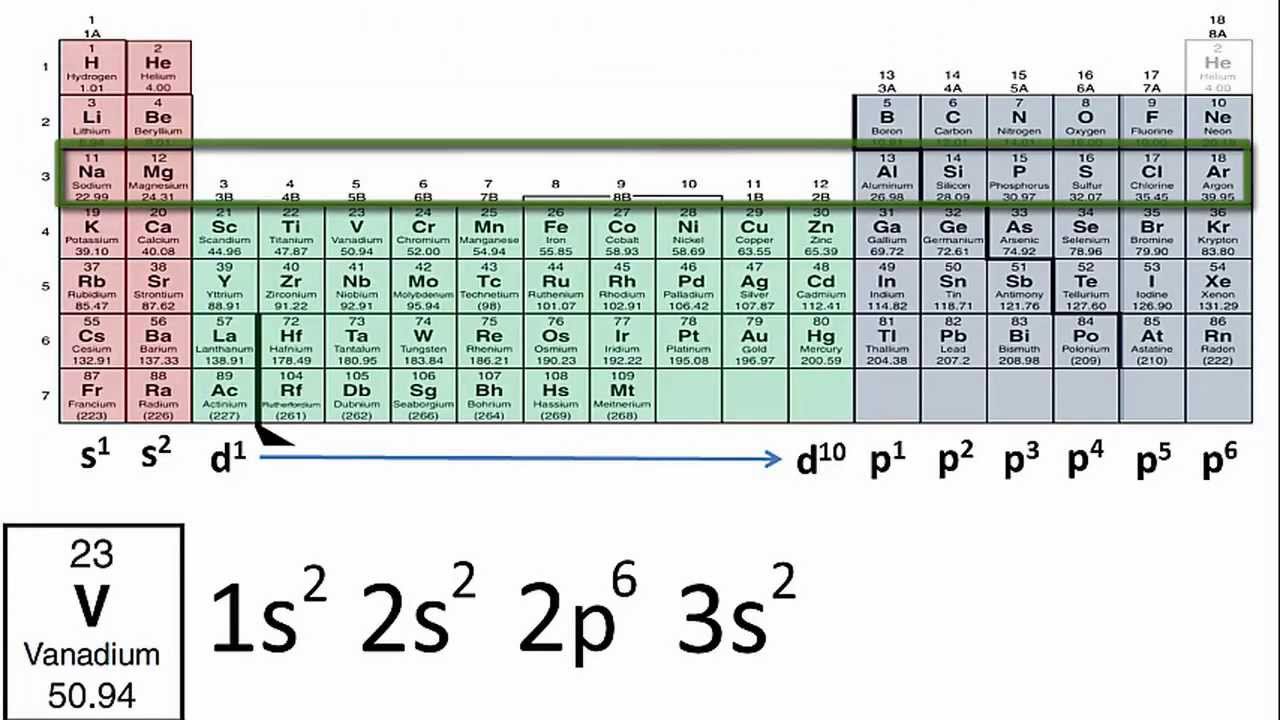
Name the element that belongs to s-block but is placed in the p-block. Uranium has 92 electrons out of which 6 valence electrons are present in the 5f3 6d1 7s2 outer orbitals of atom. This set of Chemistry Multiple Choice Questions & Answers (MCQs) focuses on Electronic Configurations and Types of Elements: s, p, d and f Blocks. 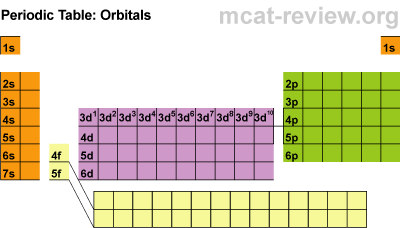
How many valence electrons does a Uranium atom have? It is located in group null and period 7 in the modern periodic table. Uranium is the 92 element on the periodic table. The colour scheme is designed to match that used by Wikipedia for its own element related articles. Each element is detailed with the name, symbol, and number of electrons in each shell. Uranium is a chemical element with the symbol U and atomic number 92. English: Diagram showing the periodic table of elements in the form of their electron shells. What is the position of Uranium in the Periodic Table? Uranium is a chemical element with symbol U and atomic number 92. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Uranium is 5f3 6d1 7s2. What is the abbreviated electronic configuration of Uranium? What is the electronic configuration of Uranium? Uranium Thermal Properties - Enthalpies and thermodynamics Optical Properties of Uranium Refractive IndexĪcoustic Properties of Uranium Speed of Sound Uranium Magnetic Properties Magnetic Type Uranium Heat and Conduction Properties Thermal Conductivity Refer to table below for the Electrical properties ofUranium Electrical Conductivity Hardness of Uranium - Tests to Measure of Hardness of Element Mohs Hardness Next we have rodeo.Refer to below table for Uranium Physical Properties Densityġ9.05 g/cm3(when liquid at m.p density is $17.3 g/cm3) 
Therefore, we can also use Krypton for it's for writing its noble gas annotation, which whose Kirton five s to 40 to Now let me move this up a little bit again. Zirconium is also located in period five dressed like strong Jim. Two p six three s to three p six for us to three d 10 four p six five s to and 40 to If we refer back to our periodic table. So it's electron configuration iss oneness to to us too. Consider the first two members of period 3: sodium (Na) and. Elements that are in the same period have chemical properties that are not all that similar. 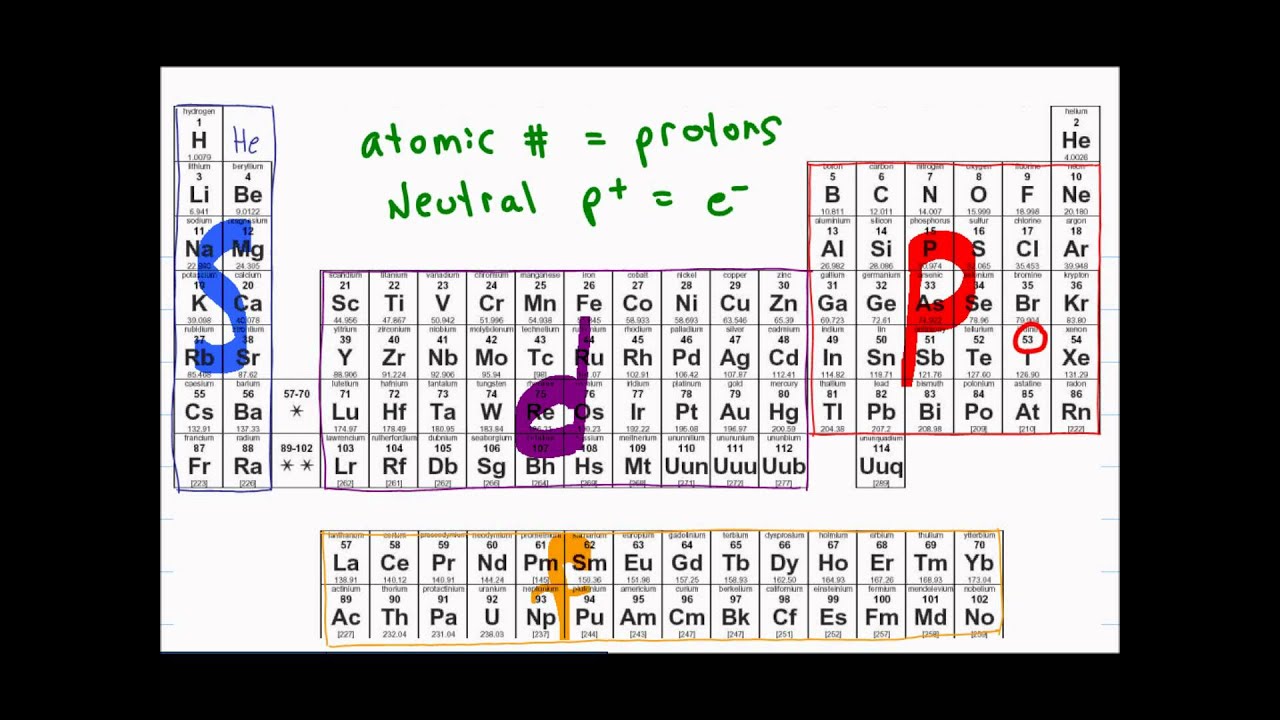
The periods are numbered 1 through 7 on the left-hand side of the table. And in five s to next up, we have zirconium, which has an atomic number of 40. In each period (horizontal row), the atomic numbers increase from left to right. Therefore, we can use this in writing Strong Tim's noble gas imitation, which is the chemical symbol for Krypton. So we may recognize from our previous problem that this electron configuration corresponds to krypton. Two p six three s to three p six for us to three d turn four p six and five s to. It's electron configuration is oneness to to us too. So let's start with strong Doom, which has an atomic number of 38. For the specific problem, you can look at the periodic table for your reference. So the problem requires us to write the electron configuration for strontium, zirconium, rodeo and tin. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
